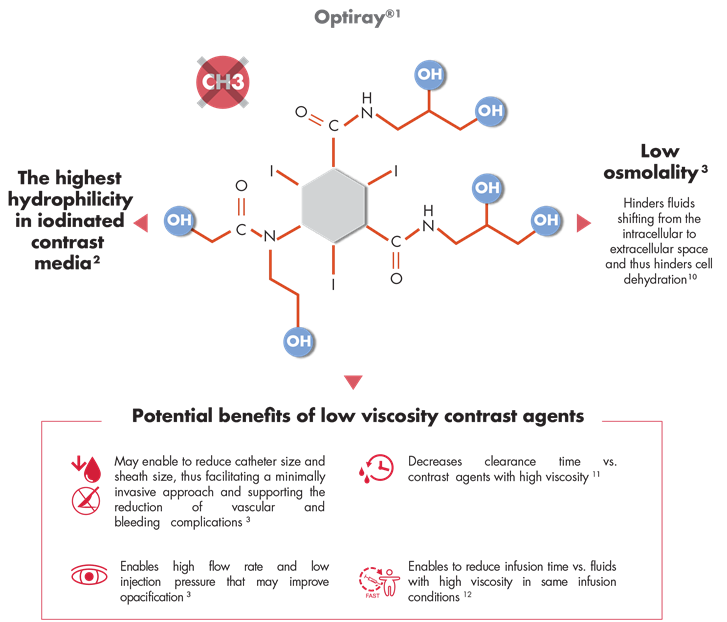
1. SmPC
2. Le Mignon MM, Azau C, Arthaud A, Bonnemain B. Preliminary
European intravenous clinical experience with a new, low
osmolar, nonionic contrast medium: ioversol (Optiray®). Eur J Radiol. 1991;13:126-133.
3. Voeltz MD, et al. The Important Properties of Contrast
Media: Focus on Viscosity. J Invasiv Cardiol 2007;19(3):1A-9A.
4. Bonnemain B, et al. New iodinated, low-osmolar contrast
media. A revised concept of hydrophilicity. Invest Radiol
1990;25(Suppl 1):S104-6.
5. Azzalini L, et al. Incidence of contrast-induced acute
kidney injury in a large cohort of all-comers undergoing
percutaneous coronary intervention: Comparison of five
contrast media. Int J Cardiol 2018;273:69–73.
6. Iyengar SS, et al. Diagnostic accuracy of high-definition
CT coronary angiography in high-risk patients. Clin Radiol
2016;71:151-158.
7. Buerke et al, Microbiologic Contamination and Time
Efficiency of Use of Automatic MDCT Injectors with Prefilled
Syringes: Results of a Clinical Investigation. AJR 2010;
194:299–303
8. Vogl TJ et al. An observational study to evaluate the
efficiency and safety of ioversol pre-filled syringes compared
with ioversol bottles in contrast-enhanced examinations. Acta
Radiol. 2012: 1-7
9. Pichler W et al. Hygiene-related considerations for
examinations conducted with contrast media. Wiener Klinisches
Magazin; March 2009:36-39
10. Thomsen HS. Management of acute adverse reactions to
contrast media. In: Thomsen HS, editor. Contrast Media: Safety
Issues and ESUR Guidelines. Heidelberg, Springer;2006. pp.
19-25
11. Jost G, et al. The Impact of the Viscosity and Osmolality
of Iodine Contrast Agents on Renal Elimination. Invest Radiol.
2010;45:255-261.
12. Geenen RW, et al. Contrast-induced nephropathy:
pharmacology, pathophysiology and prevention. Insights
Imaging. 2013;4:811-820.
Read product information
P21000538 – April 2021